|


Lab safety in the Biochemistry laboratories
Biochemistry laboratory is a place for experimentation and learning, therefor you have to take responsibility for your own personal safety and those who work with you, Incidents usually caused by negligence, but you can help to prevent it by follow the exact instructions. in addition, follow the instructions of supervisor or a laboratory technician, the following are some of the basic safety rules that will help you to protect yourself and others from exposure to injury in the laboratory.
"1. Conduct yourself in a responsible manner at all times in the laboratory.
2. Follow all written and verbal instructions carefully. If you do not understand a direction or part of a procedure, ASK YOUR TEACHER BEFORE PROCEEDING WITH THE ACTIVITY.
3. Never work alone in the laboratory. No student may work in the science classroom without the presence of the teacher.
4. When first entering a science room, do not touch any equipment, chemicals, or other materials in the laboratory area until you are instructed to do so.
5. Perform only those experiments authorized by your teacher. Carefully follow all instructions, both written and oral. Unauthorized experiments are not allowed.
6. Do not eat food, drink beverages, or chew gum in the laboratory. Do not use laboratory glassware as containers for food or beverages.
Horseplay, practical jokes and pranks are dangerous and prohibited
7. Be prepared for your work in the laboratory. Read all procedures thoroughly before entering the laboratory. Never fool around in the laboratory. Horseplay, practical jokes, and pranks are dangerous and prohibited.
8. Always work in a well-ventilated area.
9. Observe good housekeeping practices. Work areas should be kept clean and tidy at all times.
10. Be alert and proceed with caution at all times in the laboratory. Notify the teacher immediately of any unsafe conditions you observe.
11. Dispose of all chemical waste properly. Never mix chemicals in sink drains. Sinks are to be used only for water. Check with your teacher for disposal of chemicals and solutions.
12. Labels and equipment instructions must be read carefully before use. Set up and use the equipment as directed by your teacher.
13. Keep hands away from face, eyes, mouth, and body while using chemicals or lab equipment. Wash your hands with soap and water after performing all experiments.
14. Experiments must be personally monitored at all times. Do not wander around the room, distract other students, startle other students or interfere with the laboratory experiments of others.
15. Know the locations and operating procedures of all safety equipment including: first aid kit(s), and fire extinguisher. Know where the fire alarm and the exits are located.
16. Know what to do if there is a fire drill during a laboratory period; containers must be closed, and any electrical equipment turned off.
17. Any time chemicals, heat, or glassware are used, students will wear safety goggles. NO EXCEPTIONS TO THIS RULE!
18. Contact lenses may be not be worn in the laboratory.
19. Dress properly during a laboratory activity. Long hair, dangling jewelry, and loose or baggy clothing are a hazard in the laboratory. Long hair must be tied back, and dangling jewelry and baggy clothing must be secured. Shoes must completely cover the foot. No sandals allowed on lab days.
20. A lab coat or smock should be worn during laboratory experiments.
Correct protective gear must be worn in the laboratory
21. Report any accident (spill, breakage, etc.) or injury (cut, burn, etc.) to the teacher immediately, no matter how trivial it seems. Do not panic.
22. If you or your lab partner is hurt, immediately (and loudly) yell out the teacher's name to get the teacher's attention. Do not panic.
23. If a chemical should splash in your eye(s) or on your skin, immediately flush with running water for at least 20 minutes. Immediately (and loudly) yell out the teacher's name to get the teacher's attention.
24. All chemicals in the laboratory are to be considered dangerous. Avoid handling chemicals with fingers. Always use a tweezer. When making an observation, keep at least 1 foot away from the specimen. Do not taste, or smell any chemicals.
25. Check the label on all chemical bottles twice before removing any of the contents. Take only as much chemical as you need.
26. Never return unused chemicals to their original container.
27. Never remove chemicals or other materials from the laboratory area.
28. Never handle broken glass with your bare hands. Use a brush and dustpan to clean up broken glass. Place broken glass in the designated glass disposal container.
29. Examine glassware before each use. Never use chipped, cracked, or dirty glassware.
30. If you do not understand how to use a piece of equipment, ASK THE TEACHER FOR HELP!
31. Do not immerse hot glassware in cold water. The glassware may shatter.
32. Do not operate a hot plate by yourself. Take care that hair, clothing, and hands are a safe distance from the hot plate at all times. Use of hot plate is only allowed in the presence of the teacher.
33. Heated glassware remain very hot for a long time. They should be set aside in a designated place to cool, and picked up with caution. Use tongs or heat protective gloves if necessary.
34. Never look into a container that is being heated.
35. Do not place hot apparatus directly on the laboratory desk. Always use an insulated pad. Allow plenty of time for hot apparatus to cool before touching it."*
General Safety Signs in the Laboratories
|
Mandatory Signs | Señales de obligatoriedad
|

|

|

|
|
Refer to Instruction Manual/Booklet
Consulte el Manual de Instrucciones / Folleto
|
Wash Your Hands
Lava Tus Manos
|
General Mandatory Action Sign
General Obligatoria Acción Sesión
|
 |
 |

|
|
Wear Protective Clothing
Use ropa protectora
|
Wear Safety Footwear
Use Calzado de Seguridad
|
Wear Protective Gloves
Use guantes de protección
|

|

|

|
|
Wear a Mask
Use una máscara
|
Wear a Face Shield
Use una careta
|
Wear Eye Protection
Use Protección de los ojos
|
|
Warning & Hazard Signs | Advertencia y Muestras de peligro
|

|

|

|
|
Corrosive Substance
Sustancia Corrosiva
|
Toxic Material
Materiales Tóxicos
|
Harmful Substance
Sustancia Nociva
|
The guide of precautions in the laboratory

Safety tools in the laboratory

|
 |
 |
|
Lab Coat
Bata De Laboratorio
|
Lab Safety Shoes
Zapatos de seguridad Lab
|
Safety Gloves
Guantes de seguridad
|
Material Safety Data Sheet | Ficha De Datos De Seguridad De Materiales

صحيفة بيانات سلامة المادة ويرمز لها بالرمز (MSDS)، هي استمارة تحتوي على بيانات عن خصائص مادة معينة. وهي عنصر مهم في سلامة المختبرات، فهي توفر للعاملين والطلاب والباحثين والمسعفين إجراءات المعالجة والتعامل مع هذه المادة بطريقة آمنة.
ويتضمن معلومات من قبيل البيانات الفيزيائية (نقطة الانصهار، ونقطة الغليان، نقطة الوميض، إلخ)، السمية، والآثار الصحية، والإسعافات الأولية، والتفاعلية، وطريقة التخزين والتخلص منها، ومعدات الوقاية، وإجراءات التعامل معها في حالة انسكابها.
إن صحيفة بيانات السلامة للمواد MSDS هي نظام واسع الاستخدام لتصنيف المعلومات عن المواد الكيميائية، والمركبات الكيميائية، والخلائط الكيميائية. قد تحتوي البيانات على معلومات تشمل تعليمات الاستخدام الآمن والمخاطر المحتملة المرتبطة بمواد أو منتجات معينة. ويمكن العثور على هذه الصحيفة حيثما تستخدم المواد الكيميائية أو عبر الانترنت. قد تختلف الصيغة الدقيقة للصحيفة من مصدر إلى مصدر داخل البلد وفقا للطريقة المحددة في المتطلبات الوطنية.
Fire Diamond | Fuego Diamante

النظام القياسي لتحديد مخاطر المواد للاستجابة للحالات الطارئة ويرمز له بالإنجليزية NFPA 704
وهو معيار معتمد من قبل الرابطة الوطنية للحماية من الحرائق في الولايات المتحدة الأمريكية

شعار الرابطة الوطنية للحماية من الحرائق في الولايات المتحدة الأمريكية
تقسم ألماسة النار إلى أربع أقسام ملونة بألوان مختلفة تشير إلى معان مختلفة، فاللون الأزرق يشير إلى مستوى الخطر على الصحة، واللون الأحمر يشير إلى الاشتعالية، واللون الأصفر إلى التفاعلية الكيميائية، واللون الأبيض يحتوي رمزا خاصا للمخاطر الخاصة.
وتتراوح قيم الصحة والاشتعالية والتفاعلية من 0 (لا خطر، مادة عادية) إلى 4 (خطير).
Flammability (Red)
|
| 0 |
Materials that will not burn under typical fire conditions (e.g. Carbon tetrachloride), including intrinsically noncombustible materials such as concrete, stone and sand (Materials that will not burn in air when exposed to a temperature of [820 °C (1,500 °F)] for a period of 5 minutes)
|
| 1 |
Materials that require considerable preheating, under all ambient temperature conditions, before ignition and combustion can occur (e.g. mineral oil). Includes some finely divided suspended solids that do not require heating before ignition can occur. Flash point at or above 93 °C (200 °F).
|
| 2 |
Must be moderately heated or exposed to relatively high ambient temperature before ignition can occur (e.g. diesel fuel) and multiple finely divided suspended solids that do not require heating before ignition can occur. Flash point between 38 and 93 °C (100 and 200 °F).
|
| 3 |
Liquids and solids (including finely divided suspended solids) that can be ignited under almost all ambient temperature conditions (e.g. gasoline, acetone). Liquids having a flash point below 23 °C (73 °F) and having a boiling point at or above 38 °C (100 °F) or having a flash point between 23 and 38 °C (73 and 100 °F).
|
Health (Blue)
|
| 0 |
Poses no health hazard, no precautions necessary and would offer no hazard beyond that of ordinary combustible materials (e.g. wood)
|
| 1 |
Exposure would cause irritation with only minor residual injury (e.g. acetone, sodium bromate)
|
| 2 |
Intense or continued but not chronic exposure could cause temporary incapacitation or possible residual injury (e.g. diethyl ether)
|
| 3 |
Short exposure could cause serious temporary or moderate residual injury (e.g. chlorine, liquid hydrogen, carbon monoxide)
|
Instability / Reactivity (Yellow)
|
| 0 |
Normally stable, even under fire exposure conditions, and is not reactive with water (e.g. helium)
|
| 1 |
Normally stable, but can become unstable at elevated temperatures and pressures (e.g. propene)
|
| 2 |
Undergoes violent chemical change at elevated temperatures and pressures, reacts violently with water, or may form explosive mixtures with water (e.g. white phosphorus, potassium, sodium)
|
| 3 |
Capable of detonation or explosive decomposition but requires a strong initiating source, must be heated under confinement before initiation, reacts explosively with water, or will detonate if severely shocked (e.g. ammonium nitrate, chlorine trifluoride)
|
Special Notice (White)
|
OX
|
Oxidizer, allows chemicals to burn without an air supply (e.g. potassium perchlorate, ammonium nitrate, hydrogen peroxide).
|
--W--
|
Reacts with water in an unusual or dangerous manner (e.g. cesium, sodium, sulfuric acid).
|
SA
|
Simple asphyxiant gas. Specifically limited to the following gases: nitrogen, helium, neon, argon, krypton and xenon.
|
COR
ACID, ALK
|
Corrosive; strong acid or base (e.g. sulfuric acid, potassium hydroxide)
Acid or alkaline, to be more specific
|
Emergency Tools in Laboratories
Fire Types and Fire Extinguishers
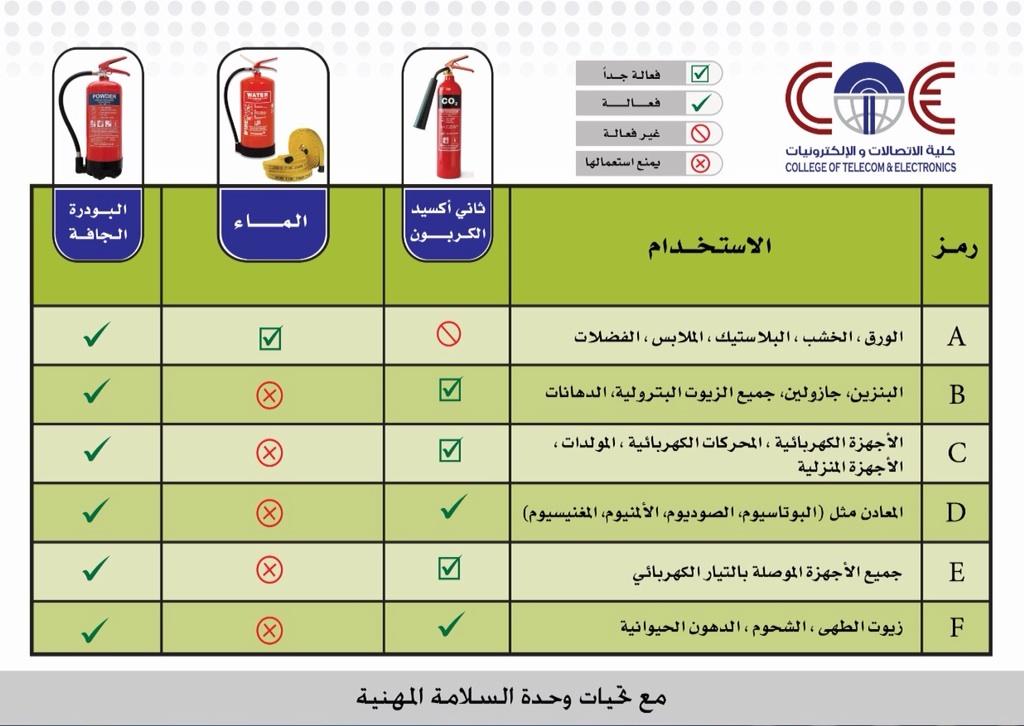
How to use the fire extinguisher

For further guidance, please visit the page of Emergency and Disaster Center (EDC) of the university

References
- nobel.scas.bcit.ca* / ISO.org / MSDS.com / NFPA.org
- All images sources is its uniform resource locator (URL) | Todas las fuentes de imágenes es su localizador uniforme de recursos (URL)

|